Cancer vaccines have been pursued for decades, yet repeated late-stage disappointments led many to doubt whether therapeutic vaccination could ever deliver meaningful clinical benefit. Those disappointments were not only about antigen choice, they were also about immune context: many early programs could prime T cells, but could not keep them functional inside an immunosuppressive tumor microenvironment. 1,2
The checkpoint era changed that context. PD-1 blockade demonstrated that many tumors are immunogenic but restrained by inhibitory signaling, and that releasing those brakes can produce durable anti-tumor activity in a subset of patients. That success reframed vaccines from stand-alone “immune starters” into potential “immune directors” that might work best when paired with agents that prevent exhaustion and restore effector function. 3, 4
Today the most productive vaccine conversations shifted from “can we induce immunity?” to “can we maintain it, broaden it, and translate it clinically in the right disease setting?”
A Short Historical Overview: What Went Wrong Before
A pivotal milestone was sipuleucel-T (PROVENGE), approved by the U.S. FDA in 2010 for metastatic castration-resistant prostate cancer.5 In the Phase III IMPACT trial, sipuleucel-T was associated with a statistically significant 22–25 % reduction in the risk of death compared with placebo (hazard ratio ~0.78; 95 % CI ~0.61–0.98; p≈0.03). Median overall survival was 25.8 months in the sipuleucel-T arm versus 21.7 months in the control arm, an improvement of 4.1 months. At 36 months, the estimated survival probability was approximately 31.7 % with sipuleucel-T versus 23.0 % with placebo. Despite this clear survival benefit, there was no significant difference in time to objective disease progression or other conventional intermediate endpoints such as PSA response, highlighting how immune kinetics can diverge from traditional measures of tumor shrinkage or progression.6
A second cautionary chapter involved shared tumor-associated antigens. In resected MAGE-A3-positive NSCLC, adjuvant MAGE-A3 immunotherapy did not improve disease-free survival in the Phase III MAGRIT trial. 1 A separate Phase III trial in melanoma (the DERMA study) evaluated the MAGE-A3 antigen-specific immunotherapeutic as adjuvant treatment in patients with completely resected stage IIIB or IIIC MAGE-A3–positive cutaneous melanoma. In this randomized, double-blind, placebo-controlled study, median disease-free survival (DFS) was nearly identical between the MAGE-A3 arm and placebo (11.0 months vs. 11.2 months; HR 1.01, 95 % CI 0.88–1.17; p = 0.86), indicating no significant DFS benefit. Median overall survival (OS) also did not differ between groups (HR 1.06, 95 % CI 0.89–1.26; p = 0.52) at final analysis. Development of the agent in melanoma was subsequently discontinued based on these null results.7
This failure is frequently cited because it underscores a recurring vulnerability of “one-size-fits-most” antigen strategies: immunogenicity alone does not guarantee durable clinical effect.
“Like the early days of immunotherapy, cancer vaccines for a time had a long history of failure… Many people felt that this was because they couldn’t be combined with anti–PD-1.”
—Dr. Eric Rubin
Now we know that early vaccine programs were not “biologically impossible”; they were often deployed in settings of high tumor burden and entrenched immune suppression, without a reliable way to rescue vaccine-primed T cells once they entered tumors. 2
Why Personalized Cancer Vaccines Are Re-Emerging in the Checkpoint Era
Early PD-1 blockade studies established safety, activity, and immune correlates of response in multiple tumor types, demonstrating that reversing inhibitory signaling can yield meaningful anti-tumor activity. 3 This created a mechanistic rationale for revisiting vaccines not as monotherapy, but as combination partners intended to expand antigen-specific breadth and depth under a checkpoint-supported immune environment. 4
In development-strategy terms, anti–PD-1 converted vaccines from “hopeful immune sparks” into plausible “immune amplifiers,” provided antigen quality and disease setting were chosen correctly.8
Personalized Neoantigen Cancer Vaccines and Precision Oncology
A major conceptual shift has been the move from shared antigens to personalized neoantigens derived from each patient’s tumor mutations.9 Two landmark first-in-human melanoma studies demonstrated feasibility and immunogenicity of personalized neoantigen vaccination using peptide and RNA platforms. 8, 9
- Ott and colleagues showed that a personalized neoantigen vaccine developed at Dana-Farber Cancer Institute, commonly referred to as NeoVax (synthetic long peptides given with the adjuvant poly-ICLC), could safely induce neoantigen-specific T-cell responses in patients with high-risk melanoma. In the corresponding first-in-human study, 4 of 6 vaccinated patients remained recurrence-free at a median follow-up of ~25 months, while the two patients who relapsed subsequently achieved complete responses after PD-1 blockade.8
- Sahin and colleagues reported that an individualized RNA “Mutanome” neoantigen vaccine developed by BionTech (RNA-LPX multi-epitope format), could induce potent neoantigen-specific immune responses in melanoma. In the Phase I study, 8 of 8 patients developed vaccine-induced immune responses, and clinical activity was observed when vaccination was combined with checkpoint inhibition, supporting the feasibility of multi-epitope RNA vaccination.9
Together, these studies made a practical case that individualized mutation sets can be translated into manufacturable vaccines that generate measurable anti-tumor immune responses.
mRNA-Based Personalized Cancer Vaccines: Merck–Moderna’s V940 Program
The most advanced modern program in personalized cancer vaccination is Moderna/Merck’s individualized neoantigen therapy mRNA-4157 (V940), developed and evaluated in combination with pembrolizumab, representing the most clinically mature example of a personalized neoantigen vaccine strategy to date. 10
In the randomized Phase 2b KEYNOTE-942 study in patients with resected high-risk melanoma, V940 plus pembrolizumab demonstrated a statistically significant improvement in recurrence-free survival compared with pembrolizumab alone, providing one of the clearest clinical signals thus far that individualized vaccination can deliver incremental benefit on top of an established anti–PD-1 backbone. Importantly, this effect was observed in the adjuvant setting, where tumor burden is minimal and immune-mediated relapse prevention is a clinically meaningful and regulatorily accepted objective. 10
According to the KEYNOTE-942 study design (ClinicalTrials.gov ID: NCT03897881), the program was intentionally structured around recurrence-free survival as a primary endpoint, aligning vaccine biology with long-term immune control rather than short-term tumor shrinkage, and situating the program within the contemporary adjuvant melanoma development framework where PD-1 inhibition is standard of care.
On the basis of the Phase 2b results, the mRNA-4157 program has advanced into Phase III evaluation (INTerpath-001), positioning it as one of the first individualized neoantigen vaccine to reach registrational-intent testing in combination with a checkpoint inhibitor. 11
In a recent Arc Nouvel interview, Eric Rubin, MD, emphasized why this approach represents a departure from earlier cancer vaccine strategies:
“That program is now in phase III. I think it’s scientifically very attractive because it’s based on each patient’s individual tumor mutations.” — Dr. Eric Rubin
From a strategic and biological perspective, the critical innovation of V940 is not mRNA as a delivery platform alone, but the systematic selection of patient-specific neoantigens derived from each individual tumor’s mutational landscape, an approach that mirrors the logic of precision oncology already applied to targeted therapies, but redirects it toward immune specificity rather than oncogenic signaling. 8, 10
Looking forward, what can reasonably be anticipated is not a universal vaccine effect, but a more refined understanding of which patients derive the greatest benefit, how neoantigen quality and clonal architecture influence outcomes, and whether durable immune memory translates into long-term reductions in recurrence and distant metastasis, questions that Phase III evaluation is now positioned to answer.
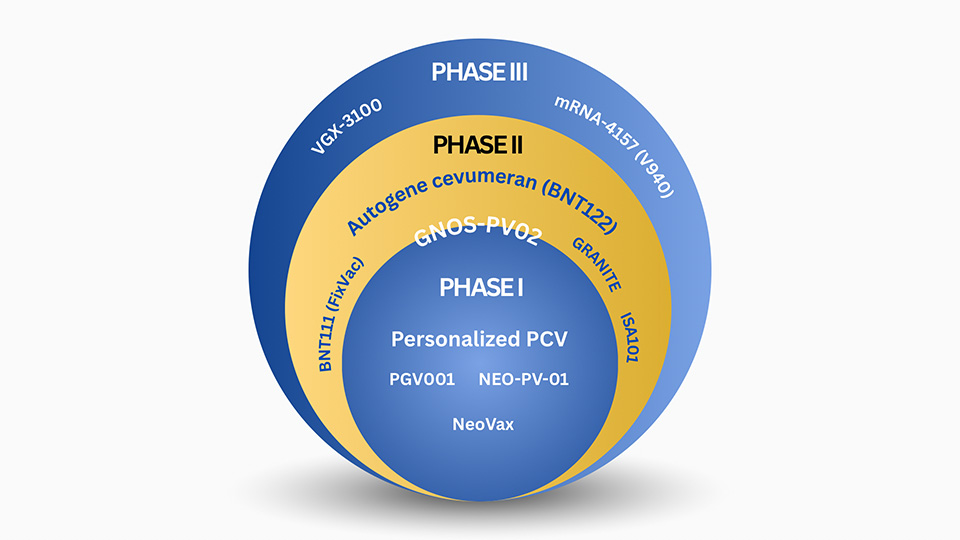
A Pipeline View
The programs highlighted below are included not because they are merely “interesting,” but because each has produced peer-reviewed clinical data that interrogates a core historical failure mode of cancer vaccines—antigen selection, immune durability, disease setting, or combination strategy. Inclusion required at least one of: antigen-specific T-cell expansion in patients, evidence of functional persistence under checkpoint blockade, and/or a clinical signal or endpoint selection consistent with immune-mediated disease control.
- Moderna/Merck — mRNA-4157 (V940) + pembrolizumab (Melanoma, INTerpath-001 (Phase III) ongoing. ClinicalTrials.gov ID: NCT05933577)
- What’s published: (5-year follow-up, Phase 2b KEYNOTE-942/mRNA-4157-P201) — At a median 5 years (pre-planned analysis), V940 + pembrolizumab showed sustained improvement in the primary endpoint (RFS), reducing risk of recurrence or death by 49% vs pembrolizumab alone (HR 0.510; 95% CI 0.294–0.887; one-sided nominal p=0.0075). INTerpath-001 (Phase III, adjuvant melanoma) is is active, not recruiting (pembro + V940 vs pembro + placebo).
- Why it matters: This is now one of the clearest long-follow-up datasets supporting the “personalized neoantigen mRNA vaccine + PD-1 backbone” strategy in the adjuvant relapse-prevention setting—raising the bar for durability expectations.
- BioNTech/Genentech/Memorial Sloan Kettering Cancer Center — autogene cevumeran (BNT122/RO7198457) in resected PDAC (adjuvant program)
- What’s published: Nature (2023) showed the adjuvant regimen (atezolizumab + autogene cevumeran + chemotherapy) induced vaccine-expanded neoantigen-specific T cells in 8/16 patients, and immune responders had delayed recurrence vs non-responders (extended follow-up reported as median RFS not reached vs 13.4 months). 13
- Why it matters: Direct human evidence that PDAC can generate vaccine-expanded neoantigen T cells with durability, challenging the “immunologically inaccessible” assumption.
- BioNTech — BNT111 (FixVac) in melanoma (RNA-LPX, early clinical)
- What’s published: In a Phase I trial (Nature), the RNA-LPX vaccine BNT111 (4 shared melanoma antigens) showed objective responses in efficacy-evaluable patients with measurable disease: 3/25 (12%) with monotherapy and 6/17 (35%) with BNT111 + anti-PD-1; at the selected combo dose, 5/10 (50%)14
- Why it matters: Provides clinical proof that an off-the-shelf, shared-antigen mRNA vaccine can induce robust tumor-specific immunity and immune-consistent objective responses in checkpoint-refractory melanoma.14
- Dana-Farber Cancer Institute — Personalized peptide neoantigen vaccine (NeoVax concept; melanoma first-in-human)
- What’s published: In the landmark first-in-human study (Nature), 6 patients with high-risk resected melanoma received a personalized peptide neoantigen vaccine. Neoantigen-specific CD4⁺ T-cell responses were detected in 6/6 patients, and CD8⁺ responses in ~4/6 patients, targeting ~60 % of vaccinated neoantigens. Clinically, 4/6 patients remained recurrence-free at a median follow-up of ~25-months, while the 2 patients who relapsed subsequently achieved complete responses to PD-1 blockade, suggesting vaccine-primed sensitivity.8
- Why it matters: establishes peptide personalization as a credible path with familiar manufacturing paradigms. 8
- Neon Therapeutics/BioNTech — NEO-PV-01 personalized peptide vaccine + anti–PD-1–based therapy (NSCLC)
- What’s published: In Phase Ib (Cancer Cell): 38 enrolled; 21 received NEO-PV-01 with carbo/pemetrexed/pembrolizumab (1L nonsq NSCLC). There were no treatment-related serious adverse events, and vaccination induced neoantigen-specific CD4⁺ and CD8⁺ T-cell responses, with immune and ctDNA features explored in relation to progression-free survival.15
- Why it matters: demonstrates that fully personalized peptide neoantigen vaccination can be integrated into first-line metastatic NSCLC alongside chemotherapy and PD-1 blockade, inducing durable, cytotoxic neoantigen-specific T-cell responses and epitope spreading (including KRAS) in a high-bar clinical setting.15
- Gritstone bio — ChAd68 + self-amplifying mRNA neoantigen vaccine + nivolumab/ipilimumab (advanced solid tumors)
- What’s published: In an interim Phase I study (Nature Medicine), Gritstone evaluated a personalized ChAd68 prime/samRNA boost neoantigen vaccine combined with nivolumab ± ipilimumab in patients with advanced solid tumors. Personalized vaccines were successfully manufactured for 15/15 patients, with 14 patients treated, and no dose-limiting toxicities reported. Neoantigen-specific T-cell responses were detected in 100% of evaluable patients (13/13), with responses predominantly CD8⁺, polyepitopic, and vaccine-induced. In the MSS colorectal cancer subgroup (n = 7), an exploratory analysis showed 3/7 patients (42.9%) alive at data cutoff, and among patients with longitudinal ctDNA monitoring who remained on therapy ≥6 months (n = 4), ctDNA reductions were observed in 3/4 (75%), including 2 complete ctDNA clearances.16
- Why it matters: this is a clean example of a prime–boost personalization strategy that achieved (1) high manufacturing feasibility, (2) CD8-skewed neoantigen responses in all evaluable patients, and (3) exploratory OS/ctDNA signals in “cold,” CPI-refractory MSS-CRC—a setting where objective responses are uncommon, so survival/ctDNA may be more informative early readouts.16
- Dana-Farber Cancer Institute/Harvard-affiliated investigators — Personalized neoantigen vaccine in high-risk resected RCC (peptide PCV; adjuvant-style)
- What’s published: In a Phase I trial (published in Nature), a peptide-based personalized neoantigen vaccine given after surgery to nine patients with high-risk, fully resected clear-cell renal cell carcinoma was safe and highly immunogenic, with T-cell responses detected in all patients and neoantigen-specific activity against key RCC driver mutations. At a median follow-up of ~40 months, none of the nine patients experienced disease recurrence. 17
- Why it matters: suggests vaccines may fit particularly well in “MRD-like” post-resection contexts where immune control can prevent relapse. 17
- Geneos Therapeutics — GNOS-PV02 DNA neoantigen vaccine + IL-12 plasmid + pembrolizumab (HCC)
- What’s published: In a Phase 1/2 trial published in Nature Medicine, the personalized DNA neoantigen vaccine GNOS-PV02 co-administered with a plasmid encoding IL-12 plus pembrolizumab was safe, feasible, and immunogenic in patients with advanced hepatocellular carcinoma. Among 36 treated patients, the objective response rate (ORR) was ~30.6 % with ~8.3 % complete responses, and neoantigen-specific T-cell responses were observed in most evaluable participants. These results suggest clinical activity in an otherwise less immunotherapy-responsive tumor context and support extending personalized vaccine strategies into such settings. 18
- Icahn School of Medicine at Mount Sinai/Genentech — PGV001- Personalized neoantigen vaccination + atezolizumab (urothelial cancer)
- What’s published: In a Phase I trial published in Nature Cancer, the combination of personalized neoantigen vaccine PGV001 with the PD-L1 inhibitor atezolizumab was feasible, safe, and induced neoantigen-specific T-cell responses in all evaluable patients. At a median follow-up of ~39 months, 3 of 4 patients treated in the adjuvant setting remained recurrence-free, and 2 of 5 patients with measurable metastatic disease achieved objective responses. 19
- Why it matters: extends the platform concept beyond PD-1 into PD-L1 combinations and bladder cancer development pathways. 19
- Inovio Pharmaceuticals — VGX-3100 DNA therapeutic vaccine (HPV16/18 CIN2/3; translational relevance to HPV cancers)
- What’s published: In a randomized, double-blind, placebo-controlled Phase 2b trial, the DNA vaccine VGX-3100 targeting HPV-16/18 E6/E7 oncoproteins induced statistically significant histopathological regression of high-grade cervical intraepithelial neoplasia (CIN2/3) to low-grade or no disease: ~48 % of VGX-3100 recipients showed lesion regression compared with ~30 % in the placebo group (difference ~18 %, p≈0.034). In addition, ~40 % of vaccinated patients achieved combined lesion regression and HPV-16/18 clearance versus ~14 % with placebo.20
- Why it matters: while “precancer” is distinct from metastatic disease, this remains one of the clearest demonstrations of therapeutic vaccination efficacy against HPV-driven pathology in a rigorous randomized design.20
- ISA Pharmaceuticals — ISA101 HPV16 peptide vaccine + nivolumab (HPV16+ cancers)
- What’s published: In a single-arm Phase II trial (JAMA Oncology; NCT02426892), ISA101 + nivolumab was evaluated in 24 patients with incurable HPV16+ cancers (mostly OPC). ORR was 33% (8/24; 2 CR, 6 PR; 90% CI 19–50%), with median DoR 10.3 months, median PFS 2.7 months, and median OS 17.5 months (with 5/8 responses ongoing at the time of analysis).20
- Why it matters: Illustrates a vaccine-as-sensitizer strategy in virally driven cancers where antigen targets (HPV E6/E7) are well-defined.20
Viewed collectively, this pipeline does not suggest that cancer vaccines are broadly effective across tumors or lines of therapy. Rather, it clarifies where they may be clinically defensible: settings with defined antigens (neoantigenic or viral), lower or controlled tumor burden, and a checkpoint backbone that preserves effector function. Importantly, several programs on this list earn inclusion not because they “worked,” but because they answered the right clinical questions, by selecting appropriate endpoints, demonstrating immune mechanism in patients, or exposing where biology still fails. From an industry perspective, this is progress: the field is no longer asking whether cancer vaccines can work in principle, but under what conditions they justify further development.
The Future: What Changes If Cancer Vaccines Succeed?
If a personalized cancer vaccine wins in Phase 3 and enters routine adjuvant use, the ripple effects could be structural:
- Oncology practice could shift “left” (earlier)
Adjuvant and MRD-like settings are already where vaccine biology appears most coherent: low tumor burden, relapse-prevention endpoints, and time for immune maturation. Success would accelerate a broader move toward immune-interception and relapse prevention rather than late-line salvage.
- “Platform thinking” becomes real in immuno-oncology
Personalized vaccines behave more like systems than single molecules: sequencing → antigen selection → manufacturing → release → delivery. Programs like autogene cevumeran explicitly track clonal T-cell expansion magnitude and persistence, and link it to recurrence outcomes, effectively making immune engineering measurable.
- Biopharma operations and supply chains change
Personalization makes manufacturing, turnaround time, chain-of-identity, and cold-chain logistics part of “clinical performance,” not just operations. A successful product will reward companies that treat CMC as a strategic competency from day one, not a late-stage compliance sprint.